Tropic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name 3-Hydroxy-2-phenylpropanoic acid | |
| Other names 2-Phenylhydracrylic acid; Tropate | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.201 |
| EC Number |
|
| KEGG |
|
| MeSH | C011377 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C9H10O3 |
| Molar mass | 166.176 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Tropic acid is a chemical with IUPAC name 3-hydroxy-2-phenylpropanoic acid and condensed structural formula HOCH2CHPhCOOH. It is a laboratory reagent used in the chemical synthesis of atropine and hyoscyamine. Tropic acid is a chiral substance, existing as either a racemic mixture or as a single enantiomer.
Synthesis
| Tropic acid synthesis 1:[1] | Tropic acid synthesis 2:[1] |
|---|---|
 | 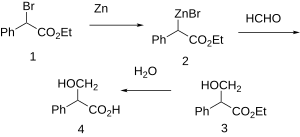 |
References
- ^ a b Gadzikowska, M; Grynkiewicz, G (2002). "Tropane alkaloids in pharmaceutical and phytochemical analysis". Acta Poloniae Pharmaceutica. 59 (2): 149–60. PMID 12365608.
- ^ Sletzinger, Paulsen, U.S. patent 2,390,278 (1945 to Merck & Co.).
- ^ Blicke, U.S. patent 2,716,650 (1955 to University of Michigan).
- ^ DE 923426, Blicke, Frederick Franklin, "Verfahren zur Herstellung von α-substituierten β-Oxypropionsäuren [Process for the preparation of α-substituted β-oxypropionic acids]", published 1955-02-14, assigned to Sterling Drug Inc.
- ^ Blicke, F. F., Raffelson, H., Barna, B. (January 1952). "The Preparation of Tropic Acid". Journal of the American Chemical Society. 74 (1): 253–253. doi:10.1021/ja01121a504.
- v
- t
- e











