Nitrosation
Nitrosation is a process of converting organic compounds into nitroso derivatives, i.e., compounds containing the R-NO functionality.
C-Nitroso compounds
C-Nitroso compounds, such as nitrosobenzene, are typically prepared by oxidation of hydroxylamines:
- RNHOH + [O] → RNO + H2O
S-Nitroso compounds
S-Nitroso compounds (S-nitrosothiols) are typically prepared by condensation of a thiol and nitrous acid:[1]
- RSH + HONO → RSNO + H2O
O-Nitroso compounds
O-Nitroso compounds are similar to S-nitroso compounds, but are less reactive because the oxygen atom is less nucleophilic than the sulfur atom. The formation of an alkyl nitrite from an alcohol and nitrous acid is a common example:
- ROH + HONO → RONO + H2O
N-Nitrosamines
N-Nitrosamines, including the carcinogenic variety, arise from the reaction of nitrite sources with amino compounds, which can happen during the curing of meat. Typically, this reaction occurs when the nucleophilic nitrogen of a secondary amine attacks the nitrogen of the electrophilic nitrosonium ion:[2]
- NO2− + 2 H+ → NO+ + H2O
- R2NH + NO+ → R2N-NO + H+
Formation of an N-nitrosamine:
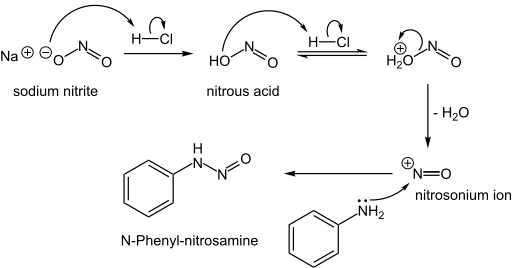
The nitrosamine can then lose water through protonation to form diazonium cation, which is a very useful intermediate to form different compounds.
References
- ^ Wang, P. G.; Xian, M.; Tang, X.; Wu, X.; Wen, Z.; Cai, T.; Janczuk, A. J. (2002). "Nitric Oxide Donors: Chemical Activities and Biological Applications". Chemical Reviews. 102 (4): 1091–1134. doi:10.1021/cr000040l. PMID 11942788.
- ^ López-Rodríguez, Rocío; McManus, James A.; Murphy, Natasha S.; Ott, Martin A.; Burns, Michael J. (2020-09-18). "Pathways for N -Nitroso Compound Formation: Secondary Amines and Beyond". Organic Process Research & Development. 24 (9): 1558–1585. doi:10.1021/acs.oprd.0c00323. ISSN 1083-6160. S2CID 225483602.
External links
- Nitrosation of Amines
- v
- t
- e
- Nitroxyl anion (NO−; oxonitrate(1-), hyponitrite anion)
- Nitric oxide (NO⋅; nitrogen monoxide)
- Nitrosonium (NO+; nitrosyl cation)
| sGC |
|
|---|
(prodrugs)
- Nitrates: Diethylene glycol dinitrate (DEGDN)
- Erythritol tetranitrate (ETN)
- Ethylene glycol dinitrate (EGDN; nitroglycol)
- Isosorbide mononitrate (ISMN)
- Isosorbide dinitrate (ISDN)
- Itramin tosilate
- Mannitol hexanitrate
- Naproxcinod (nitronaproxen; AZD-3582, HCT-3012)
- NCX-466
- NCX-2216
- NCX-4016
- NCX 4040
- NCX-4215
- Nicorandil
- Nipradilol (K-351)
- Nitrate (NO−
3) - Nitroatorvastatin (NCX-6560)
- Nitroflurbiprofen (HCT-1026)
- Nitrofluvastatin
- Nitroglycerin (glyceryl trinitrate (GTN))
- Nitropravastatin (NCX-6550)
- Pentaerithrityl tetranitrate (PETN)
- Propatylnitrate
- Propylene glycol dinitrate (PGDN)
- Sodium trioxodinitrate (Angeli's salt)
- Tenitramine
- Trolnitrate
- Nitroso compounds/nitrites: Nitrite (NO−
2); O-Nitroso compounds (alkyl nitrites): Amyl nitrite (isoamyl nitrite, isopentyl nitrite) - Cyclohexyl nitrite
- Ethyl nitrite
- Hexyl nitrite
- Isobutyl nitrite (2-methylpropyl nitrite)
- Isopropyl nitrite
- Methyl nitrite
- n-Butyl nitrite
- Pentyl nitrite
- tert-Butyl nitrite; S-Nitroso compounds (thionitrites): LA810
- S-Nitrosoalbumin (SNALB)
- S-Nitrosated AR545C
- S-Nitroso-N-acetylcysteine (SNAC)
- S-Nitroso-N-acetylpenicillamine (SNAP)
- S-Nitroso-N-valerylpenicillamine (SNVP)
- S-Nitrosocaptopril (SNO-Cap)
- S-Nitrosocysteine (SNC, CysNO, SNO-Cys)
- S-Nitrosodiclofenac
- S-Nitrosoglutathione (GSNO, SNOG)
- SNO-t-PA
- SNO-vWF; N-Nitroso compounds (e.g., nitrosamines): SIN-1A
- Nitrosyl compounds: Metal nitrosyl complexes: Roussin's black salt
- Roussin's red salt
- Sodium nitroprusside (SNP)
- NONOates (diazeniumdiolates): Diethylamine/NO (DEA/NO)
- Diethylenetriamine/NO (DETA/NO)
- GLO/NO
- JS-K
- Methylamine hexamethylene methylamine/NO (MAHMA/NO)
- PROLI/NO
- Spermine/NO (SPER/NO)
- V-PYRRO/NO
- Heterocyclic compounds: Furoxans: Furoxan
- REC15/2739; Sydnonimines: Feprosidnine
- Linsidomine (SIN-1)
- Molsidomine (SIN-10)
- Sydnonimine
- Unsorted: Cimlanod
- FK-409
- FR144220
- FR146881
- N-Acetyl-N-acetoxy-4-chlorobenzenesulfonamide
(inhibitors)
| NOS |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Arginase |
| ||||||||
| CAMK |
|
- Precursors: L-Arginine
- Nω-Hydroxy-L-arginine (NOHA)
- Indirect/downstream NO modulators: ACE inhibitors/AT-II receptor antagonists (e.g., captopril, losartan)
- ETB receptor antagonists (e.g., bosentan)
- L-Type calcium channel blockers (e.g., dihydropyridines: nifedipine)
- Nebivolol (beta blocker)
- PDE5 inhibitors (e.g., sildenafil)
- non-selective PDE inhibitors (e.g., caffeine)
- PDE9 inhibitors (e.g., paraxanthine)
- cGMP preferring PDE inhibitors (e.g., sildenafil, paraxanthine, tadalafil)
- Statins (e.g., simvastatin)












